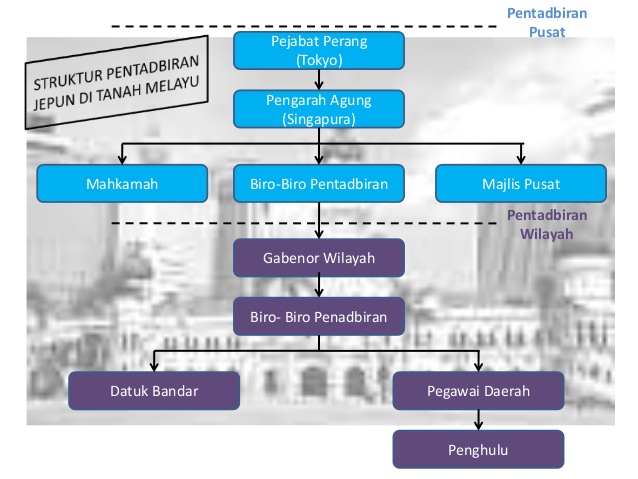
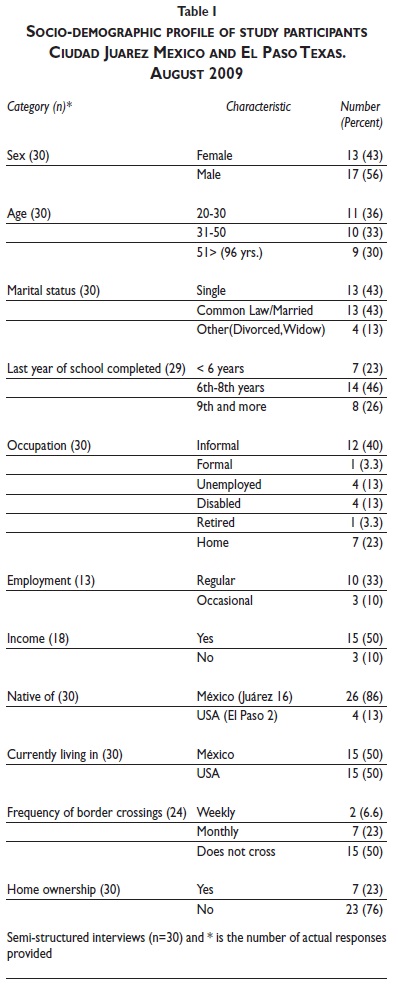
Formulas of Ionic Compounds - ThoughtCo.
How to Write Chemical Formulas Correctly. A chemical formula is something like a recipe that contains different ingredients and makes an item. For example, Cocoa Butter, Chocolate Liquor, Sugar, Lecithin and a flavoring agent makes a delicious item called Chocolate. Similarly, a compound is made of several elements and an element is made of atoms.To write chemical formulas, acquaint yourself with chemical symbols, most easily found on the periodic table of elements. The periodic table is a chart of all the known elements, and it often includes both the full name of each element and its symbol, such as H for hydrogen or Cl for chlorine.Naming and Formula Writing Overview. Learning to name and write formulas for chemical compounds requires practice with immediate feedback to help you learn from mistakes. 1. Your first step is to learn how to tell what type of compound you have. This is something you need to master before naming or writing chemical formulas.
The formula of a compound in which the combining ratio is Aluminum (Atomic symbol Al): Fluorine (atomic symbol F), 1:3 should be determined. Concept Introduction: A compound is characterized by the formula. In a compound, elements are combined in a fixed ratio.Forgetting to follow this rule is one of the most common mistakes that students make when writing formulas for ionic compounds. The empirical formula for an ionic compound indicates the smallest whole number ratio of cations and anions needed to produce an electrically neutral compound. The empirical formula is written with the cation first.

How to Write Chemical Formulas for Molecular Compounds. Formula writing for molecular compounds is probably the easiest type of formula writing. The key is memorizing the prefixes. Many of the prefixes you'll be familiar with since they are part of words we frequently use. To be successful writing formulas for molecular compounds, spend some.